Patients seeking advanced Lung Cancer Treatment in Bangalore or specialized Thoracic Cancer Treatment in Bangalore require highly precise diagnostic evaluations before initiating therapy. Similarly, successful Stomach Cancer Treatment in Bangalore and comprehensive Gastric Cancer Treatment in Bangalore depend entirely on identifying the specific molecular characteristics of the tumor.
As a premier Cancer Treatment Hospital in Bangalore and recognized as the Best Cancer Hospital in Bangalore, Dasappa Cancer Hospital utilizes advanced genomic profiling to dictate these critical medical protocols.
When navigating these complex oncological pathways, patients frequently ask what a biomarker test is and how this specific laboratory analysis alters their pharmaceutical intervention. Historically, clinical oncology relied heavily on broad-spectrum chemotherapy protocols based solely on the anatomical origin of the tumor. Modern medical oncology has shifted definitively toward precision medicine.
This advanced approach focuses on the unique genetic mutations and specific cellular proteins driving the malignant growth within an individual patient.
Understanding the clinical significance of this targeted molecular approach requires examining three specific dimensions of modern genomic diagnostics:
1. The Diagnostic Definition: Defining the exact cellular components, protein expressions, and genetic alterations identified during high-resolution molecular sequencing.
2. The Biological Mechanism: Explaining how these specific cellular mutations drive uncontrolled tumor proliferation and how identifying them enables highly targeted pharmaceutical disruption.
3. The Clinical Application: Outlining the strict necessity of molecular profiling for specific pulmonary and gastrointestinal malignancies to authorize advanced immunotherapy and targeted therapies.
Understanding the profound clinical value of precision molecular diagnostics, patients can approach their oncological care with a clear comprehension of their customized treatment trajectory.
What Is a Biomarker Test?
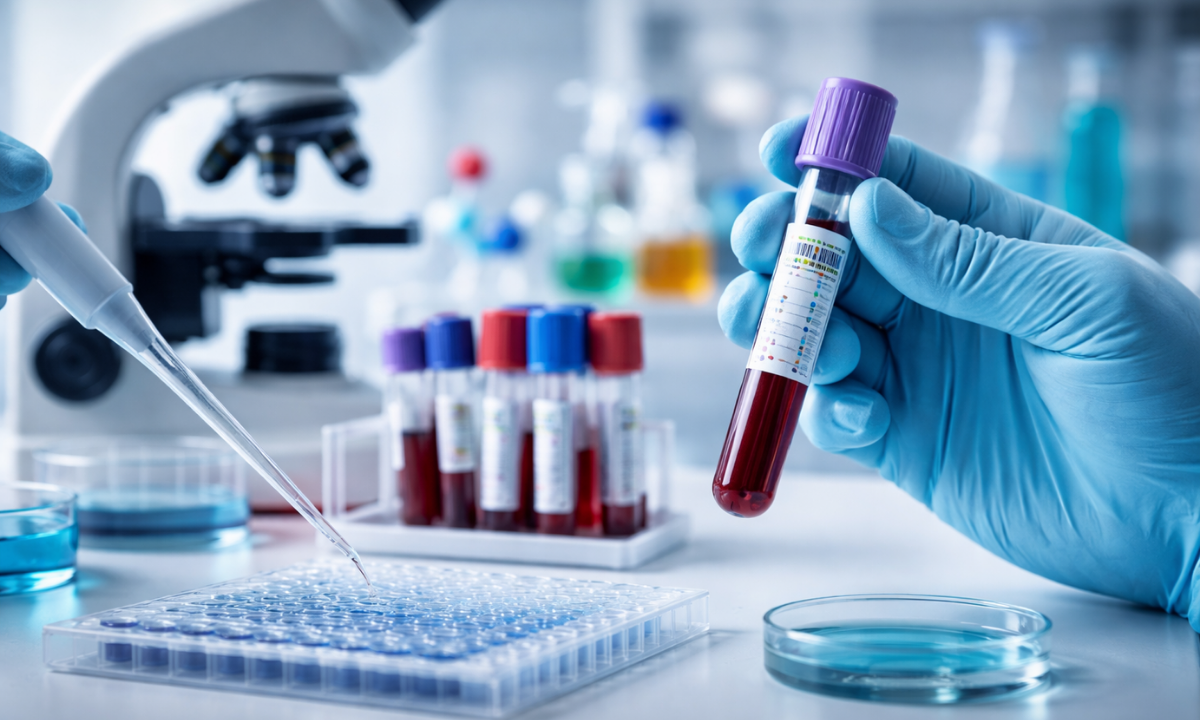
When patients and their families ask exactly what a biomarker test is, the clinical answer involves advanced molecular biology. In medical oncology, a biomarker (biological marker) is a specific, measurable molecule found within the blood, circulating bodily fluids, or directly within the malignant tumor tissue.
These markers strictly include mutated genes, overexpressed cellular proteins, or other highly specific genetic abnormalities that indicate the precise biological behavior of a malignancy.
The diagnostic process of identifying these molecules is clinically referred to as genomic profiling or tumor sequencing.
Unlike standard anatomical biopsies that only confirm the physical presence of malignant cells under a microscope, a comprehensive biomarker test for cancer thoroughly analyzes the intracellular DNA and RNA.
Clinical pathologists utilize these advanced laboratory evaluations to identify three primary categories of molecular data:
1. Genetic Mutations: Identifying highly specific alterations in the DNA sequence that directly cause the cell to divide uncontrollably.
2. Protein Overexpression: Detecting an abnormally high concentration of specific cellular receptors on the surface of the tumor cell that actively accelerate rapid tumor growth.
3. Gene Fusions: Locating specific areas where two distinct genes have physically joined together to form a new, abnormal genomic structure that directly promotes cancer development.
Mapping this exact molecular architecture, the multidisciplinary oncology team transitions from a generalized treatment model to a highly personalized medical strategy. The resulting diagnostic data directly informs the physician whether the specific tumor will respond to targeted pharmaceutical interventions or advanced immunotherapies.
This genomic precision strictly prevents the administration of ineffective toxic treatments and optimizes the patient’s overall clinical outcome.
How Biomarkers Work
To comprehend the diagnostic value of molecular sequencing, patients frequently ask exactly how biomarkers work at the cellular level. Healthy cellular replication is strictly governed by precise genetic sequences.
A malignancy develops when these internal DNA sequences undergo specific pathological alterations. These acquired mutations force the cell to proliferate uncontrollably while simultaneously developing the biological ability to evade the body’s natural immune system.
Medical oncologists categorize these pathological markers based on their precise functional role within the tumor cell.
Identifying these exact biological mechanisms is strictly mandatory prior to prescribing targeted pharmaceutical therapies.
Cellular Mechanisms of Oncogenic Biomarkers
| Biomarker Classification | Pathological Cellular Mechanism | Clinical Therapeutic Objective |
|---|---|---|
| Activating Gene Mutations | An acquired permanent alteration in the DNA sequence forces the internal cellular machinery to generate continuous, unregulated growth signals. | To administer specific small molecule inhibitors that enter the tumor cell and physically block the abnormal signaling pathway. |
| Protein Overexpression | The malignant cell produces an abnormally high density of specific surface receptors, significantly accelerating the rate of cellular division. | To utilize specialized monoclonal antibodies that attach directly to the exterior receptors, thereby cutting off all external growth stimuli. |
| Chromosomal Translocations | Two entirely separate genetic sequences break apart and fuse together inappropriately, creating a novel oncogenic protein that drives tumor formation. | To deploy highly targeted kinase inhibitors designed specifically to neutralize the resulting abnormal fusion protein. |
| Immune Evasion Expressions | The tumor cell expresses specific surface proteins that physically bind to and deactivate the patient’s attacking immune T cells. | To utilize advanced immune checkpoint inhibitors that block this deceptive protein, allowing the patient’s immune system to recognize and destroy the malignancy. |
Identifying these precise pathological drivers is the absolute foundation of precision oncology.
Rather than deploying broad systemic toxins that damage healthy tissue, oncologists utilize this highly specific molecular data to select pharmaceutical agents engineered strictly to disrupt the exact biological mechanism fueling the tumor.
Biomarker Testing in Lung cancer
In the specialized field of thoracic oncology, the medical management of Lung cancer has been completely redefined by genomic sequencing.
Specifically, for patients diagnosed with advanced non-small cell lung carcinoma, performing a comprehensive molecular evaluation is no longer considered an optional diagnostic step; it is a strict clinical mandate before initiating any pharmaceutical treatment.
The multidisciplinary tumor board strictly relies on this molecular data to evaluate the presence of several highly specific oncogenic drivers. The most critical genomic alterations evaluated within pulmonary tissue include:
1. EGFR Mutations: Alterations in the epidermal growth factor receptor gene directly cause rapid cellular division in a significant percentage of pulmonary tumors.
2. ALK and ROS1 Rearrangements: These specific chromosomal abnormalities create abnormal fusion proteins that continuously signal the tumor to grow.
3. KRAS Mutations: Identifying this specific genetic variation helps oncologists precisely predict which tumors will be highly resistant to standard targeted therapies, thereby altering the entire treatment strategy.
If the laboratory analysis successfully identifies an EGFR, ALK, or ROS1 mutation, the oncologist will immediately bypass traditional systemic intravenous chemotherapy.
Instead, the physician will prescribe a specific oral tyrosine kinase inhibitor. This is a highly targeted pharmaceutical agent engineered exclusively to enter the tumor cell and physically shut down that exact mutated signaling pathway.
Furthermore, evaluating the programmed death ligand 1 (PD-L1) protein expression is absolutely mandatory for determining a patient’s eligibility for advanced immunotherapy.
Tumors exhibiting high concentrations of the PD-L1 surface protein are highly susceptible to immune checkpoint inhibitors.
Identifying this specific biomarker, the oncologist can deploy medications that strip away the tumor’s chemical camouflage, allowing the patient’s own immune system to actively recognize and destroy the malignant cells.
Biomarker Testing in Gastric Cancer
The clinical management of stomach malignancies relies heavily on precision diagnostics. Integrating biomarker testing in gastric cancer is a mandatory protocol for determining the most effective pharmaceutical and surgical interventions.
As gastrointestinal tumors exhibit highly variable biological behaviors, treating them based strictly on their anatomical location is clinically insufficient.
Oncologists systematically evaluate the malignant gastric tissue for several specific molecular expressions to guide the treatment trajectory:
1. HER2 Overexpression: A significant percentage of gastric tumors exhibit an abnormal amplification of the human epidermal growth factor receptor 2 (HER2) protein.
Identifying this specific overexpression dictates the immediate utilization of targeted monoclonal antibodies. These specialized pharmaceutical agents specifically neutralize the HER2 receptors, effectively halting rapid cellular proliferation when combined with standard chemotherapy protocols.
2. PD-L1 Expression: Evaluating the programmed death ligand 1 status is strictly required to determine patient eligibility for advanced immunotherapy.
Identifying high concentrations of this protein allows oncologists to deploy immune checkpoint inhibitors that prevent the tumor from deactivating the patient’s immune system.
3. Microsatellite Instability (MSI) Status: Tumors classified as MSI-High possess a severe genetic inability to repair internal DNA replication errors.
This specific molecular classification highly predicts a robust clinical response to immunotherapy, allowing the medical team to prioritize these advanced treatments over highly toxic conventional chemotherapy.
Securing this precise genomic data, the multidisciplinary tumor board avoids administering ineffective, broad-spectrum treatments.
Instead, the oncology team strictly administers the exact therapeutic agents engineered to neutralize the patient’s specific gastrointestinal malignancy, thereby maximizing the statistical probability of a positive clinical outcome.
Why Choose Dasappa Cancer Hospital
We recognize that the effective medical management of complex malignancies strictly requires absolute diagnostic precision.
Our clinical infrastructure is specifically engineered to deliver advanced genomic profiling directly to our patients, ensuring that every treatment decision is grounded in concrete molecular data.
Our multidisciplinary tumor board, comprising specialized medical oncologists, surgical oncologists, and clinical pathologists, relies on state-of-the-art molecular diagnostics to formulate highly individualized treatment protocols.
Integrating comprehensive biomarker testing into our standard diagnostic pathway, we ensure several distinct clinical advantages:
1. Rapid Genomic Sequencing: We prioritize the immediate identification of highly specific genetic mutations and protein over expressions following a definitive cancer diagnosis.
This rapid molecular analysis prevents the dangerous delay of administering generalized, potentially ineffective cytotoxic chemotherapy.
2. Expert Clinical Interpretation: High-resolution genomic data requires highly specialized medical interpretation.
Our clinical team possesses the strict oncological expertise required to analyze complex tumor profiles and seamlessly translate that laboratory data into an actionable, highly targeted pharmaceutical strategy.
3. Access to Advanced Therapeutics: By definitively identifying specific oncogenic drivers, our facility immediately authorizes and administers the latest generation of targeted tyrosine kinase inhibitors and novel immune checkpoint inhibitors, maximizing the statistical probability of tumor regression.
Choosing Dasappa Cancer Hospital means securing access to a highly sophisticated, research-driven medical facility where genomic precision strictly dictates every aspect of the pharmaceutical and surgical intervention.
Conclusion
The integration of biomarker testing into standard clinical oncology represents a monumental shift in the medical management of malignant disease.
Transitioning from a broad anatomical approach to a highly specific molecular strategy, oncologists can deploy targeted therapies that precisely disrupt the exact biological mechanisms driving tumor growth.
A comprehensive genomic evaluation is no longer an optional assessment; it is the absolute, mandatory foundation of modern cancer survival. If you require a highly specialized oncological evaluation or are seeking a definitive second medical opinion regarding an advanced malignancy, contact Dasappa Cancer Hospital today.
Schedule a consultation with our multidisciplinary tumor board to secure the precise molecular diagnostics required to optimize your overall clinical outcome.