Receiving a potential lung cancer diagnosis requires immediate, highly specialized medical evaluation.
Patients seeking comprehensive Lung Cancer Treatment in Bangalore must understand that an accurate initial assessment is the absolute foundation of any effective curative strategy. Selecting an advanced Cancer Hospital in Bangalore ensures access to the precise radiological and pathological infrastructure required for this complex process.
As the Best Cancer Hospital in Bangalore, Dasappa Cancer Hospital prioritizes rapid, highly accurate diagnostic evaluations to minimize patient anxiety and accelerate the initiation of targeted oncological therapies.
As lung tissue is situated deep within the thoracic cavity, direct physical examination of a suspected tumor is anatomically impossible without specialized imaging and invasive extraction techniques.
Therefore, the diagnostic journey is a structured, multi phase clinical process. This rigorous pathway is specifically designed to identify the presence of malignant cells, determine the exact histological subtype of the tumor, and establish the precise anatomical stage of the disease prior to therapeutic intervention.
To prepare patients for this comprehensive clinical evaluation, this guide will systematically detail the exact sequence of standard diagnostic protocols, focusing specifically on the following critical phases:
- Advanced Radiological Imaging: We will explain the specific clinical utility of Low Dose Computed Tomography and Positron Emission Tomography in identifying, measuring, and mapping suspicious pulmonary nodules.
- Pathological Tissue Extraction: We will detail the standard surgical and non surgical biopsy procedures, including advanced bronchoscopy and image guided needle aspirations, which are strictly required to definitively confirm malignancy.
- Molecular Profiling and Surveillance: We will outline the integration of modern genomic blood testing and the ongoing clinical monitoring required to continuously evaluate disease progression and treatment efficacy.
Understanding the precise biological objectives of each specific test, patients can navigate this complex diagnostic pathway with greater clinical clarity and confidence in their medical team.
Advanced Imaging Modalities
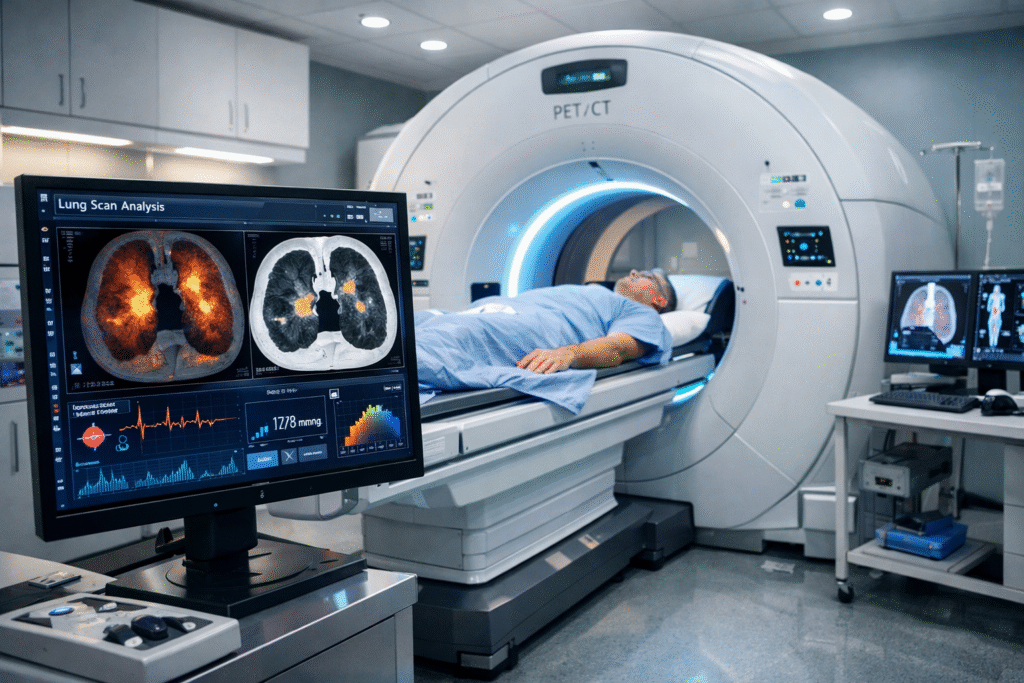
When a patient presents with specific respiratory symptoms or high risk clinical factors, the primary medical objective is to visualize the internal pulmonary architecture.
The standard chest radiograph frequently lacks the spatial resolution required to detect early stage cellular changes or minute anatomical abnormalities. Therefore, oncologists initiate a highly specific sequence of tests for lung cancer diagnosis, beginning strictly with advanced radiological imaging.
The initial phase of the diagnostic evaluation relies on two distinct imaging modalities. Each scan serves a specific clinical purpose, providing the multidisciplinary tumor board with both anatomical and biological data regarding the suspected malignancy.
Standard Radiological Diagnostic Modalities
Low Dose Computed Tomography (LDCT)
This is the definitive foundational imaging test. An LDCT scanner utilizes highly calibrated X ray beams rotating rapidly around the patient to generate highly detailed, three dimensional cross sectional images of the entire chest cavity.
The primary clinical utility of this scan is strictly anatomical. It allows the radiologist to identify, precisely measure, and map the exact spatial location of any abnormal tissue growths, clinically referred to as pulmonary nodules or masses.
Positron Emission Tomography and Computed Tomography (PET-CT)
While an LDCT scan provides exact structural dimensions, it cannot definitively verify if an identified nodule is actively malignant or merely benign scar tissue.
To evaluate the biological behavior of the mass, the medical team utilizes a PET scan. Prior to the procedure, the patient receives an intravenous injection of a specially formulated radioactive glucose compound known as fluorodeoxyglucose.
As malignant lung cancer cells undergo rapid cellular division, they possess an exceptionally high metabolic rate and aggressively consume this glucose tracer. The scanner detects the localized radiation, distinctly illuminating areas of hypermetabolic cellular activity.
Systematically combining the precise anatomical mapping of the CT scan with the metabolic activity data from the PET scan, the oncology team can highly accurately differentiate a potential malignancy from a benign granuloma or a localized pulmonary infection.
This critical radiological data does not officially confirm cancer. However, it provides the exact anatomical coordinates necessary to safely perform the next mandatory step in the diagnostic sequence: physically extracting a tissue sample from the hypermetabolic nodule.
The Definitive Biopsy Standard
While advanced radiological imaging accurately maps the physical dimensions and metabolic activity of a pulmonary nodule, it cannot provide a definitive cancer diagnosis on its own.
The absolute clinical standard for confirming a malignancy is the histological and cytological examination of the tissue itself.
To achieve this, the oncology team must perform a biopsy. This procedure involves physically extracting a sample of the abnormal cells for direct microscopic evaluation by a specialized pathologist.
The specific medical procedure utilized to extract this tissue depends entirely on the exact anatomical location, the physical size, and the accessibility of the nodule identified during the initial Computed Tomography scan.
Thoracic surgeons and interventional pulmonologists select the least invasive modality capable of securing a highly viable, uncontaminated tissue sample. The multidisciplinary team relies on several precise extraction techniques based on the specific presentation of the suspected malignancy.
Standard Pulmonary Biopsy Modalities
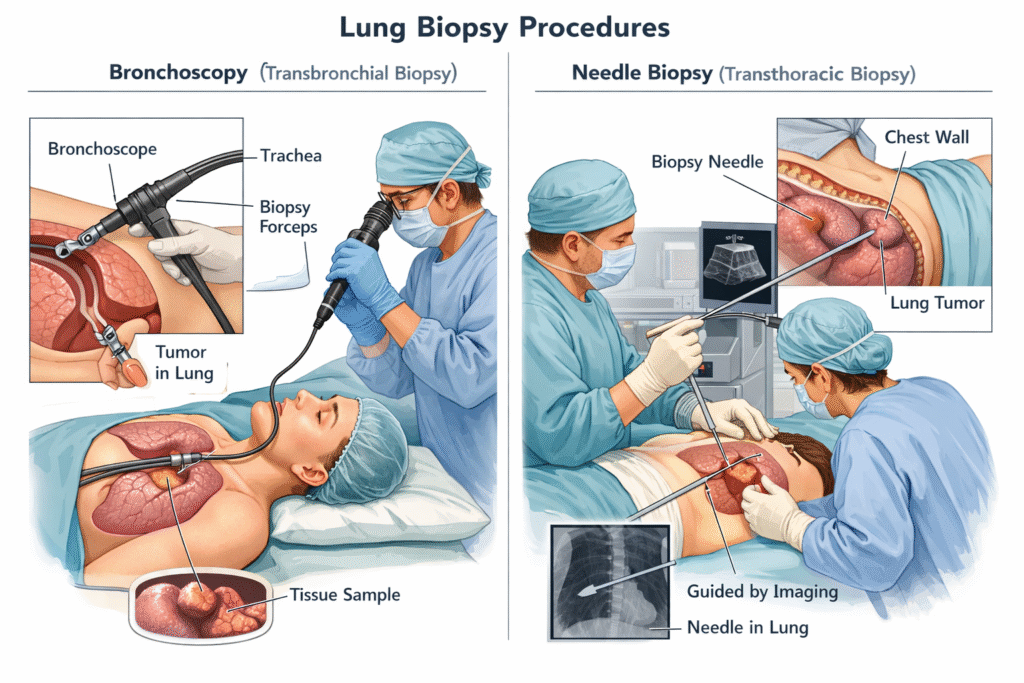
| Biopsy Procedure | Target Anatomical Location | Clinical Methodology |
| Bronchoscopy | Central airways, primary bronchi, and the trachea. | A flexible fiberoptic tube equipped with a high resolution camera is navigated through the mouth or nose directly into the primary lung passages. This allows the physician to visually inspect the mucosal lining and utilize micro forceps to extract tissue from superficially exposed tumors. |
| Endobronchial Ultrasound (EBUS) | Mediastinal lymph nodes and deeper central pulmonary tissues. | An advanced bronchoscope equipped with a localized high frequency ultrasound transducer is utilized. The ultrasound safely visualizes dense structures lying beyond the airway walls, allowing for real time, guided needle aspiration of adjacent, potentially malignant lymph nodes. |
| CT-Guided Transthoracic Needle Biopsy | Peripheral lung nodules located adjacent to the external chest wall. | An interventional radiologist utilizes continuous, real time Computed Tomography imaging to safely guide a hollow biopsy needle directly through the external chest wall, avoiding major blood vessels, straight into the target lesion to extract the required cellular material. |
| Video Assisted Thoracoscopic Surgery (VATS) | Complex, deeply embedded, or highly inaccessible pulmonary nodules. | A minimally invasive surgical procedure requiring general anesthesia. A thoracic surgeon utilizes small thoracic incisions and a specialized endoscopic camera to completely excise the suspicious nodule for immediate, comprehensive pathological review. |
Once the medical team successfully extracts the necessary cellular material, the sample is immediately transferred to the clinical pathology laboratory. The pathologist processes the tissue, applies specific chemical stains, and evaluates the cellular architecture under extreme magnification.
This exact microscopic analysis provides the definitive confirmation of the presence of lung cancer. Furthermore, it strictly dictates the specific histological subtype of the disease, categorizing the malignancy as either Non Small Cell Lung Cancer or Small Cell Lung Cancer.
Identifying this exact cellular classification is a mandatory prerequisite before the medical board can safely authorize any systemic pharmacological treatments or targeted radiation protocols.
Genomic Profiling and Liquid Biopsies
Following the physical extraction and standard microscopic confirmation of malignant cells, the diagnostic evaluation transitions into the highly advanced realm of molecular pathology.
Modern thoracic oncology no longer relies exclusively on the anatomical origin or the cellular shape of the tumor. Instead, the current clinical standard mandates comprehensive genomic profiling to identify the exact genetic mutations actively driving the rapid cellular division.
Pathologists utilize a sophisticated laboratory technique known as Next Generation Sequencing on the extracted tumor tissue.
This intricate process completely maps the DNA architecture of the malignant cells, specifically searching for actionable genetic alterations. Identifying mutations in specific genes, such as the Epidermal Growth Factor Receptor or Anaplastic Lymphoma Kinase, is a strictly mandatory diagnostic step.
The presence or absence of these precise genetic markers directly dictates whether a patient is a viable candidate for highly specific, targeted pharmacological therapies rather than standard systemic chemotherapy regimens.
In specific clinical scenarios where a traditional tissue biopsy yields insufficient cellular material or the anatomical location of the tumor presents extreme surgical risk, oncologists utilize advanced peripheral blood evaluations.
These specialized early cancer detection blood tests, clinically referred to as liquid biopsies, represent a major technological advancement in diagnostic oncology.
As malignant tumors grow within the thoracic cavity, they continuously shed microscopic fragments of their genetic material directly into the patient’s systemic circulation. By drawing a standard peripheral venous blood sample, the clinical laboratory can utilize highly sensitive assay equipment to isolate and meticulously analyze this cell free circulating tumor DNA.
While currently utilized primarily to identify specific actionable mutations when traditional tissue samples are inadequate, these advanced blood tests provide unprecedented, minimally invasive diagnostic access.
They allow the multidisciplinary medical team to safely identify the genetic profile of the tumor and promptly detect acquired pharmacological resistance without subjecting the patient to repeated, highly invasive surgical biopsy procedures.
Disease Staging and Long Term Surveillance
Once the multidisciplinary oncology team confirms the exact histological and genetic profile of the malignancy, the diagnostic phase immediately transitions into formal clinical staging.
Staging is the precise medical process of determining the total anatomical extent of the disease within the body. This comprehensive assessment is strictly required to formulate an effective, highly personalized oncological treatment strategy.
To accurately categorize the severity of the malignancy, oncologists universally utilize the TNM classification system.
This standardized medical framework evaluates three highly specific anatomical parameters based entirely on the data collected during the initial radiological scans and tissue biopsies.
The TNM Clinical Staging Framework
| Classification Parameter | Clinical Definition | Primary Diagnostic Focus |
| T (Tumor) | Quantifies the primary lesion. | Determines the exact physical size of the original pulmonary tumor in centimeters and evaluates if the mass has physically invaded adjacent structural tissues, such as the chest wall or the primary airway. |
| N (Node) | Evaluates regional lymphatic involvement. | Assesses whether the malignant cells have successfully migrated into the localized lymph nodes situated within the lung cavity or the central mediastinum. |
| M (Metastasis) | Identifies distant systemic spread. | Confirms whether the cancer has biologically metastasized beyond the thoracic cavity, establishing secondary tumors in distant anatomical locations such as the brain, liver, or skeletal system. |
By synthesizing these three exact variables, the medical board assigns a definitive clinical stage, ranging from Stage I (highly localized disease) to Stage IV (advanced metastatic disease).
This precise staging directly dictates whether the patient requires immediate surgical resection, localized radiation therapy, or systemic pharmacological interventions.
Following the establishment of the clinical stage and the initiation of targeted therapies, the patient enters a mandatory phase of continuous cancer diagnosis and monitoring. The initial diagnostic evaluation is not a singular, isolated event.
It establishes the critical biological baseline against which all future therapeutic efficacy is continuously measured.
Standard Protocols for Clinical Surveillance
Routine Restaging Scans: The medical team schedules periodic Computed Tomography and Positron Emission Tomography scans at specific intervals throughout the treatment timeline.
These scans physically quantify tumor shrinkage, confirm disease stability, or identify any anatomical signs of disease progression.
Serial Biomarker Evaluations: For patients receiving targeted pharmacological therapies, oncologists utilize ongoing liquid biopsies and advanced blood tests to track the specific levels of circulating tumor DNA.
This provides rapid, molecular level detection of potential drug resistance long before physical symptoms manifest.
Post Treatment Pulmonary Function Testing: The respiratory therapy team continuously evaluates the patient’s objective lung capacity to ensure they are safely tolerating the ongoing treatments without sustaining severe, irreversible structural damage to the healthy pulmonary tissues.
Maintaining a rigorous, highly structured surveillance schedule, the oncology team ensures that the treatment protocol remains precisely optimized for the patient’s dynamic biological response to therapy.
Why Choose Dasappa Cancer Hospital? Expert Diagnostic Infrastructure at Dasappa Cancer Hospital
Navigating a complex pulmonary evaluation requires a highly coordinated medical approach. Delays in imaging or tissue analysis can significantly increase patient anxiety and postpone the initiation of critical therapies.
Dasappa Cancer Hospital provides an elite clinical infrastructure specifically engineered to accelerate the diagnostic timeline without compromising pathological accuracy.
Under the expert medical leadership of Dr. Pandu Dasappa, a highly distinguished surgical oncologist with over twenty seven years of clinical experience, our 75 bed super specialty facility consolidates all necessary diagnostic technologies entirely under one roof.
Our dedicated thoracic oncology program features several distinct clinical advantages:
- Advanced Thoracic Radiology: Our on site imaging department features the latest generation Low Dose Computed Tomography scanners and high resolution Positron Emission Tomography equipment, guaranteeing immediate access to precise anatomical mapping.
- Minimally Invasive Biopsy Suites: Our interventional pulmonology team routinely performs Endobronchial Ultrasound and precise CT guided needle aspirations, safely extracting critical tissue samples while minimizing surgical risk and patient discomfort.
- In House Molecular Pathology: Our advanced clinical laboratory is fully equipped to perform rapid Next Generation Sequencing and liquid biopsies. This allows our oncologists to identify actionable genetic mutations and initiate targeted pharmacological therapies weeks faster than standard external laboratories.
- Multidisciplinary Tumor Board Review: Every single diagnostic case is rigorously evaluated by a collaborative panel of thoracic surgeons, medical oncologists, radiologists, and specialized pathologists to ensure the final clinical staging and subsequent treatment plan are absolutely flawless.
Prioritizing rapid, highly coordinated diagnostic evaluations, our medical team ensures that every patient transitions from their initial consultation to an active, personalized treatment phase with optimal speed and clinical precision.
Conclusion
While undergoing a comprehensive pulmonary evaluation is undoubtedly stressful, it is the mandatory first step toward a curative outcome.
A precise lung cancer diagnosis is not merely about identifying the presence of a tumor. It is a highly structured clinical process designed to map the exact biological and genetic profile of the malignancy. By fully understanding the specific sequence of radiological scans, tissue biopsies, and molecular blood tests, patients can actively participate in their medical journey with confidence.
If you are experiencing persistent respiratory symptoms or require a second opinion regarding a recent pulmonary screening, contact Dasappa Cancer Hospital today to schedule a comprehensive diagnostic evaluation with our thoracic oncology specialists.